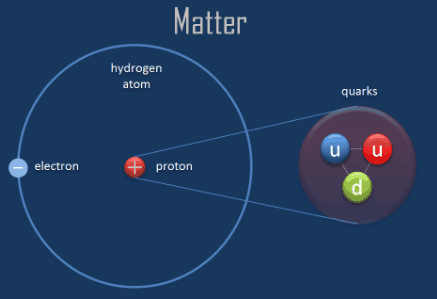
A water molecule is electrically neutral since it forms covalent bond, then how can it exert a force of attraction on hydroPhilic head of soap molecules? - Quora
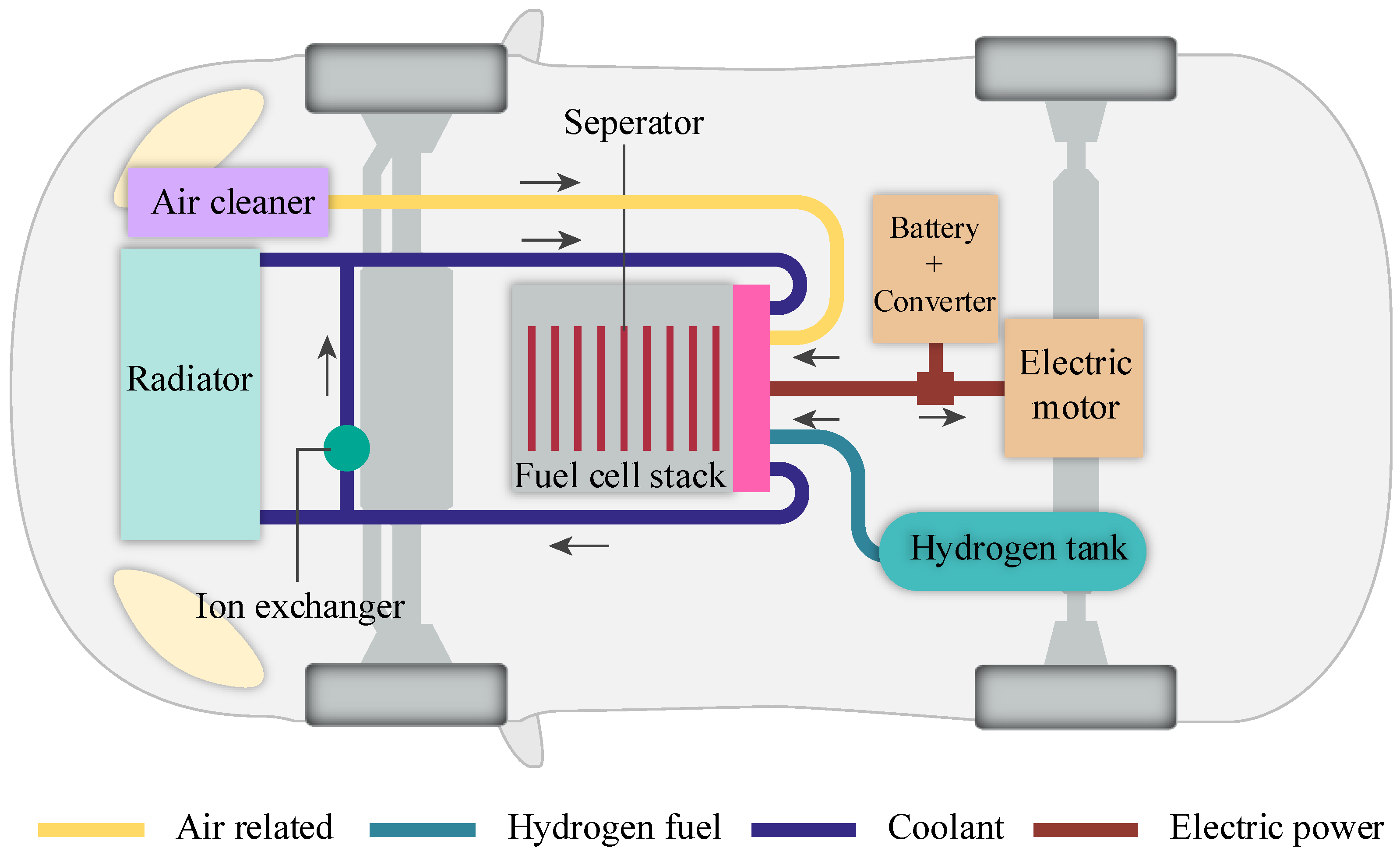
Applied Sciences | Free Full-Text | Portable Prototype of Hydrogen Fuel Cells for Educational Training

Find the atomic polarizability of Hydrogen atom Using QM Model for the Electron Cloud Pr 4-2 - YouTube